Issue Management Software
Manage deviations, nonconformances, complaints, CAPAs, and investigations all in one flexible, compliant system built for life sciences.
Absolute control over every quality event—with data integrity baked in.
Design your issue management processes your way
Use our truly configurable workflows to build the exact steps, required fields, permissions, and signature points for your unique deviations, CAPAs, investigations and more. No IT help required.
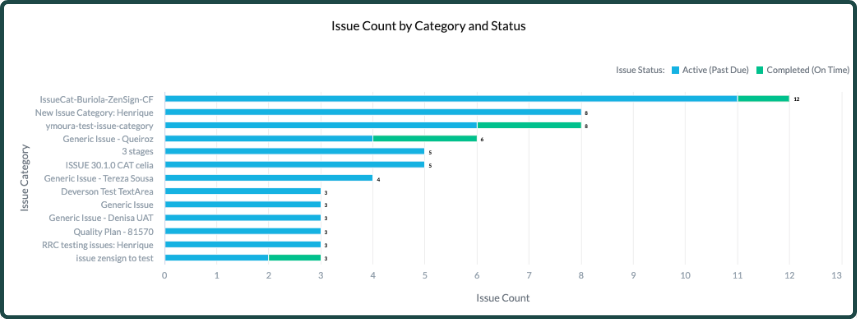
Categorize your issues – and your data – to reflect how you actually operate
Structure issue types, root causes, severity levels, and product or site associations in ways that make sense for your organization. Then, use these categories to filter your data and spot critical patterns.

Protect data integrity with the right access
Set granular permissions that control who can view, edit, and approve issue stages by role, team, and location.
Additional features in the ZenQMS Issues module that Quality teams love:
- Easily search and find any issue
- Wide variety of custom fields to structure your issue data collection in an organized way
- Build follow-up tasks directly into your issue management process
- Full, visible, searchable, and exportable Audit Trail records that regulators understand
- Built in due dates to keep your team on track
- Assign detailed permissions for issue access and escalation
- Email notifications to alert your team of upcoming or past due items
- Export any item to PDF
- APIs to connect to your other systems
More ZenQMS Modules
Training
Never guess the status of your team's training again. Just check the dashboard.
Documents
Don't chase another signature. Automatically notify your team.
Audits
Organize audits from planning to execution so you're always audit-ready.
Insights
Uncover the trends and data that can fuel continuous improvement.
Add-Ons
Get the most out of ZenQMS with powerful add-on features.
